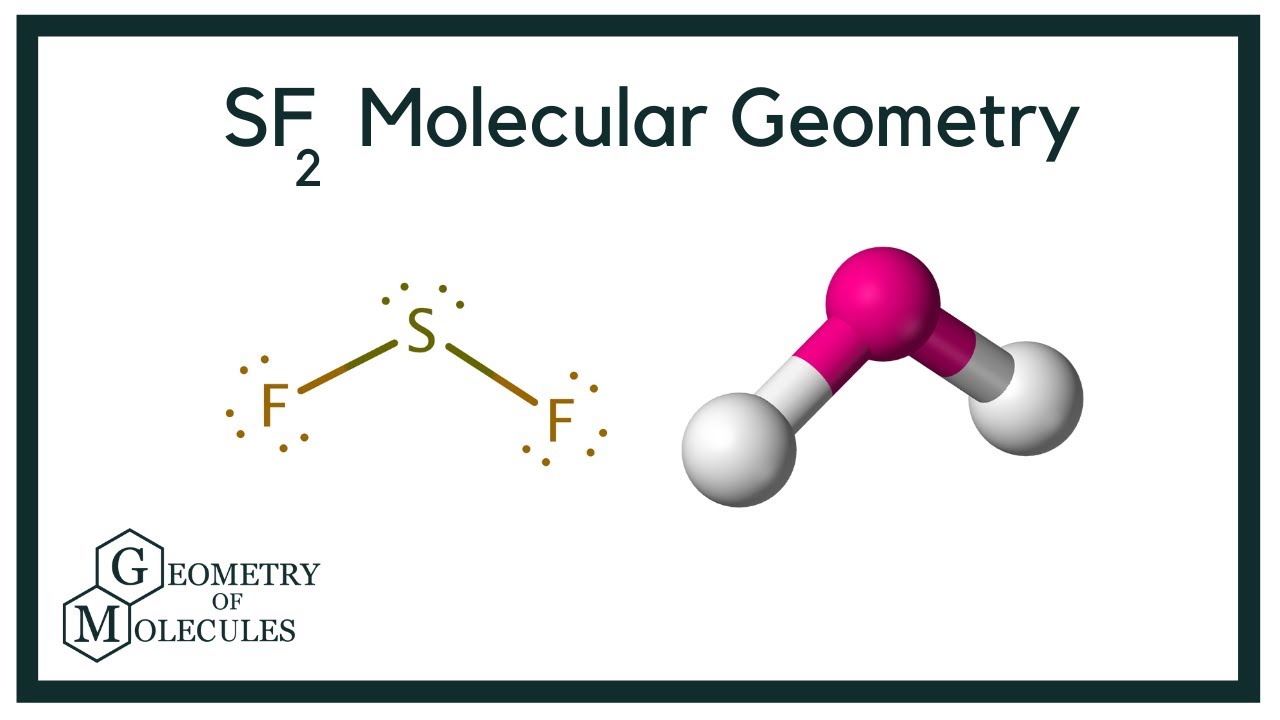
When one s orbital and 3 p orbitals sign up with together to form the SF2 molecular orbital, sp3 hybridization of SF2 molecule is formed. Atoms in the routine table are classified as complies with: SF2 particle is made of one sulfur, two fluorine atoms. The facility sulfur atom of SF2 has two only pairs of electrons, resulting in tetrahedral SF2 electron geometry. The initial step is to illustration molecular geometry of SF2 molecule, to determine lone sets of electrons in the central sulfur atom 2nd action is to determine SF2 hybridization, and the third action is to provide ideal symbols for SF2 molecular geometry.  
A three-step strategy for drawing SF2 molecular can be made use of.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
